Please see attached file. I'm a little stuc


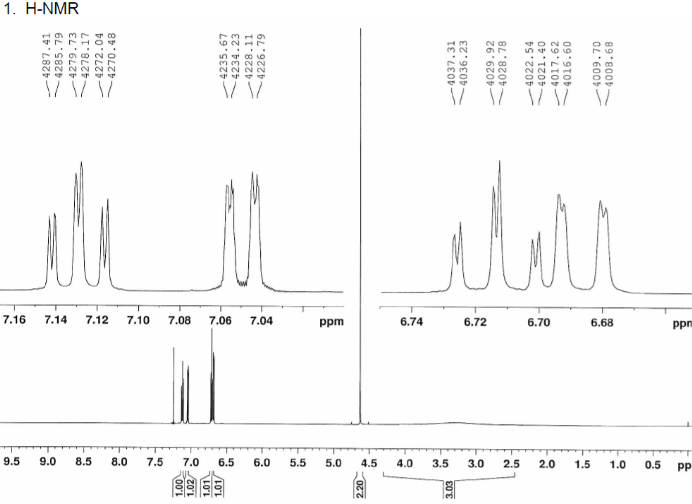
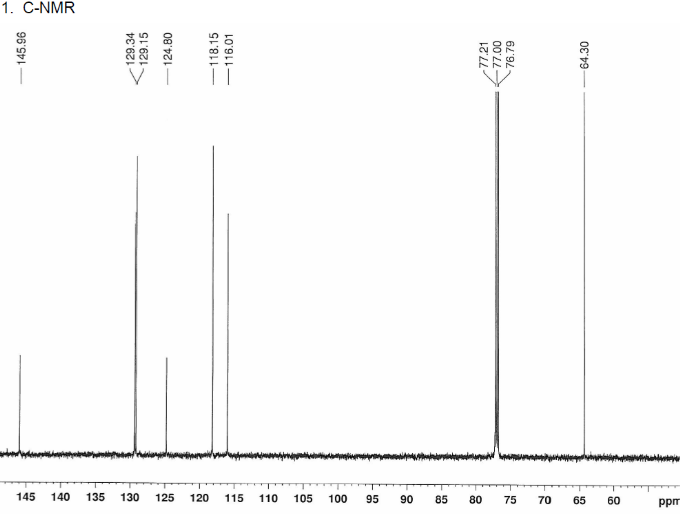




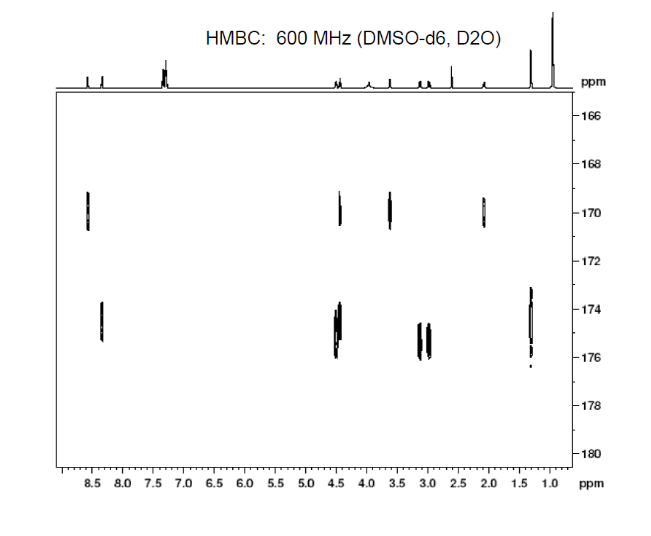
2D.
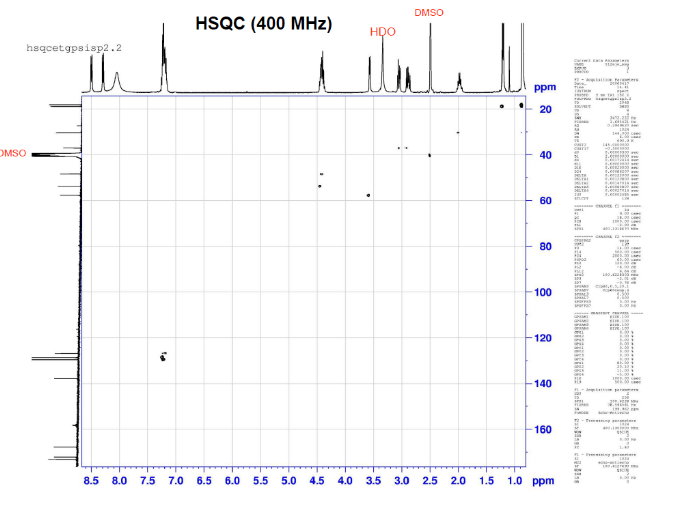
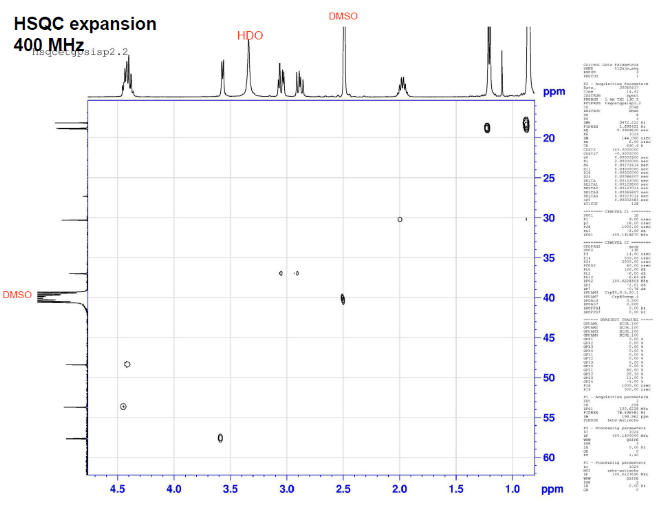
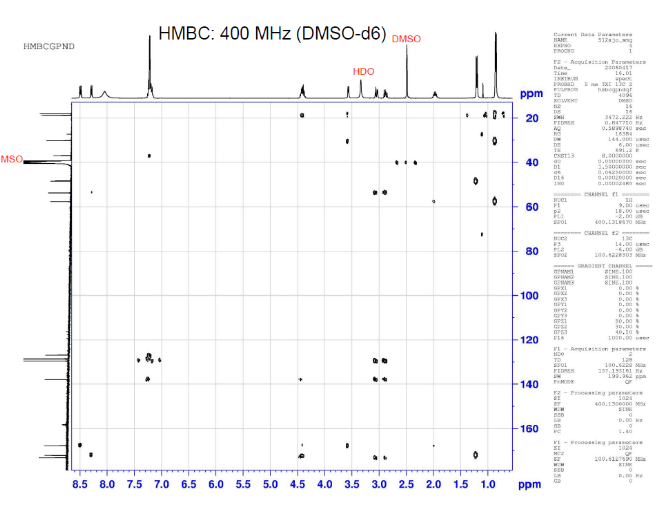
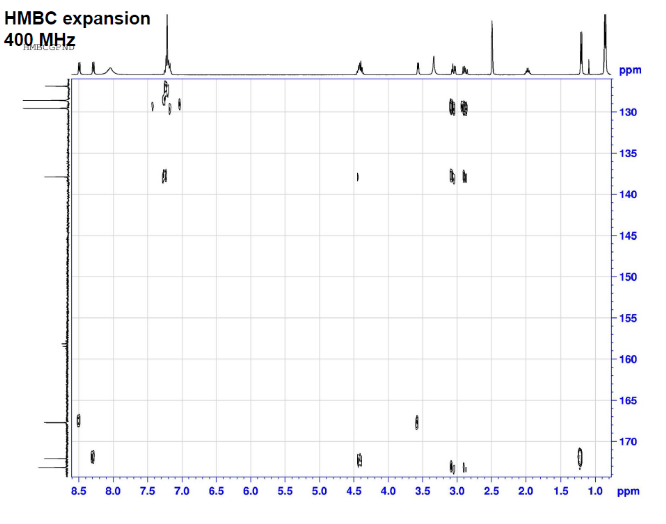
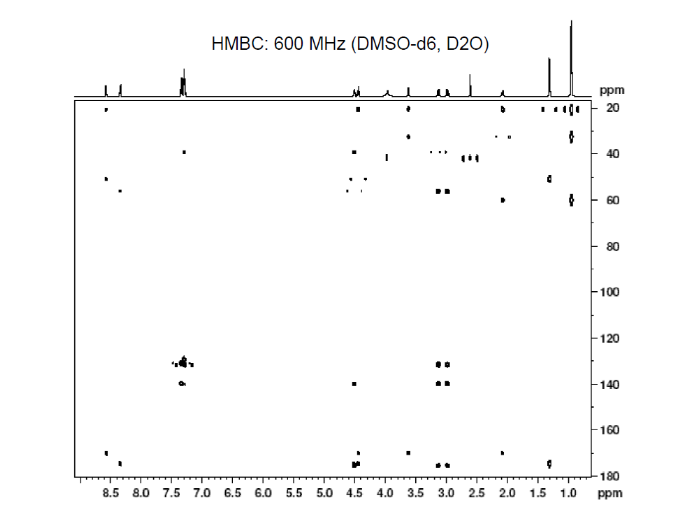
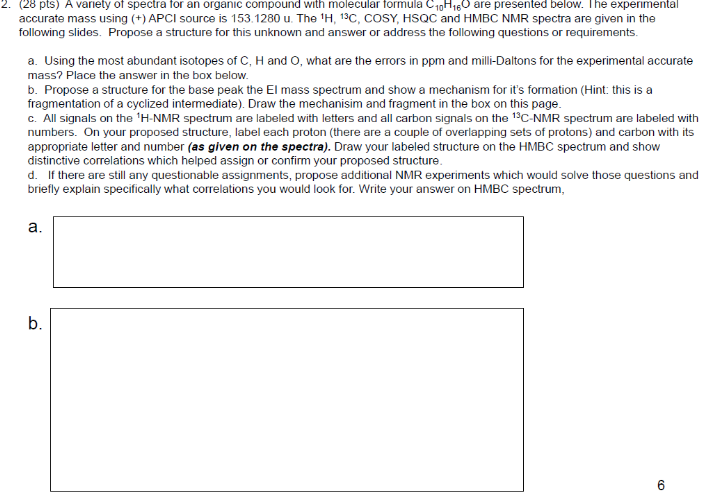
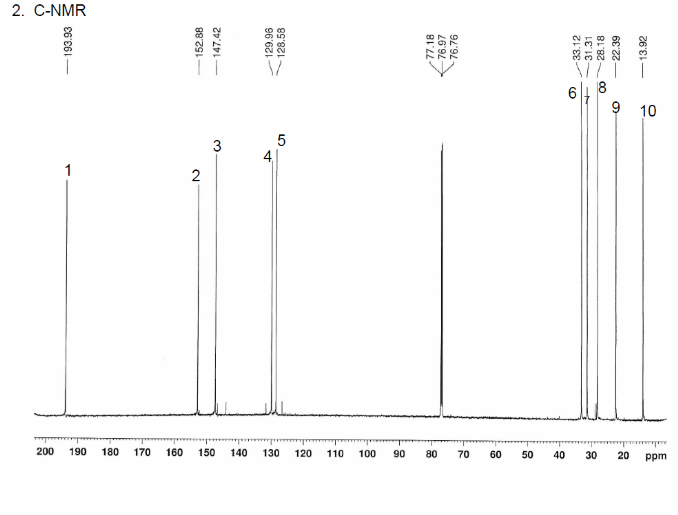
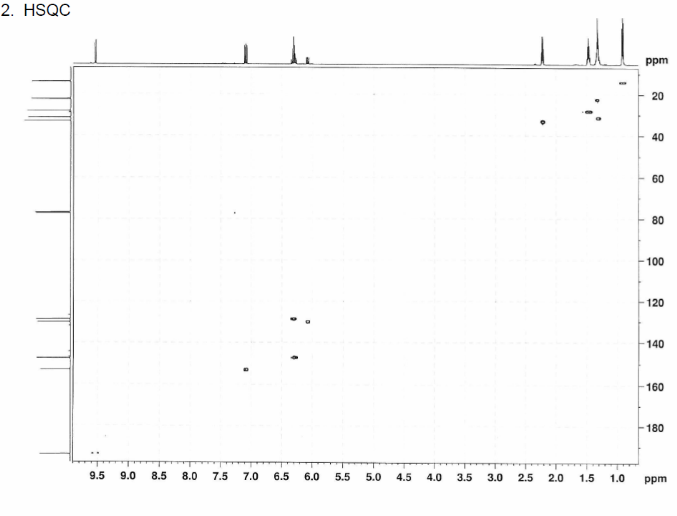
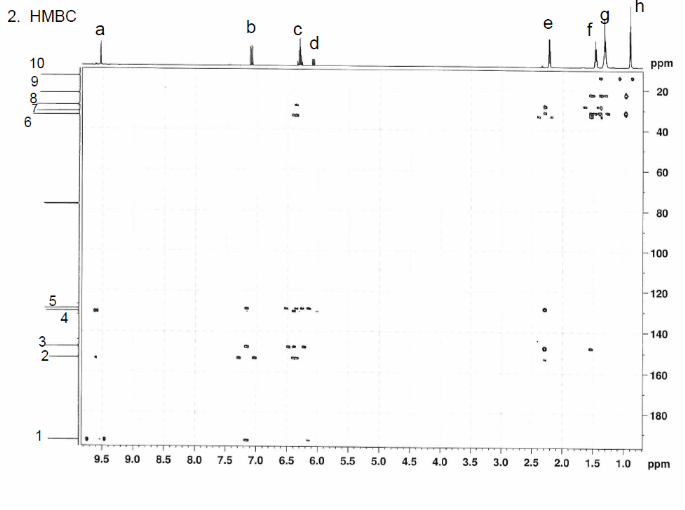
This is proton detected experiment shows carbons are 2-3 bonds away from protons as absolute value. Small couplings arising from long range interaction.3 bond coupling are larger than 2bond couplings. HMBC shows 2,3 bond; C,H
all proton detected experiments. We would like to do more experiments like qualitative and quantitative analysis or qualitative analysis, analysing composition of substance and purity of compound. We can also learn and understand how number of double bonds /rings can be worked out from molecular formula. We can also try to find Index of hydrogen deficiency. help identify compounds and determining structure is first goal of course in this study and experimentations.
Most importantly is trying to understand Deduction of information about structural features of a compound from percentage composition data, MS, 1H-NMR or IR.
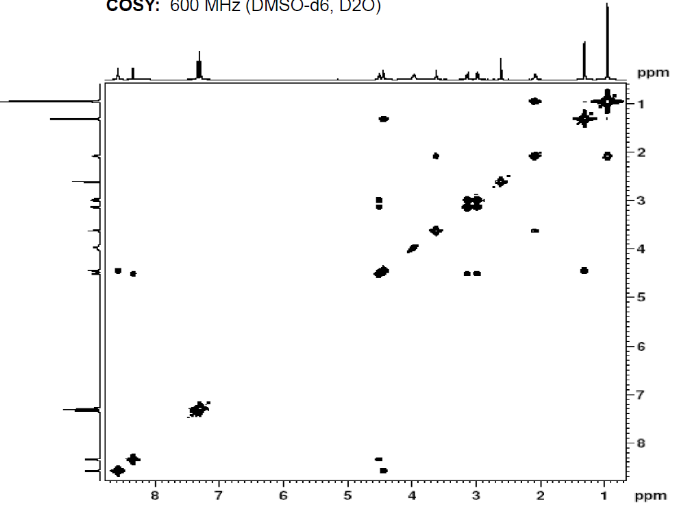
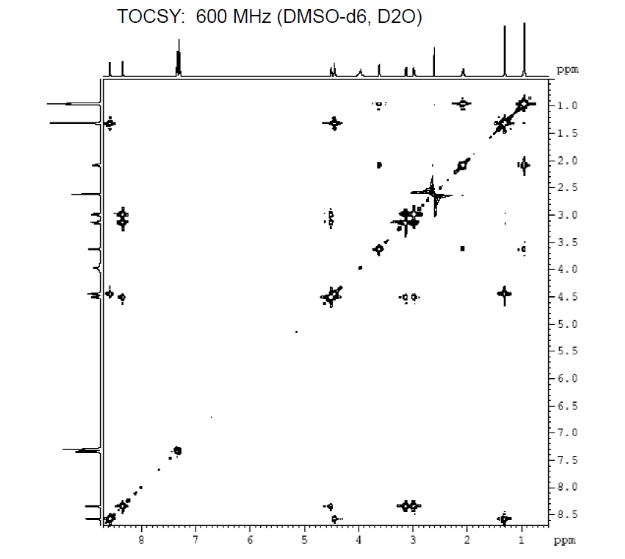
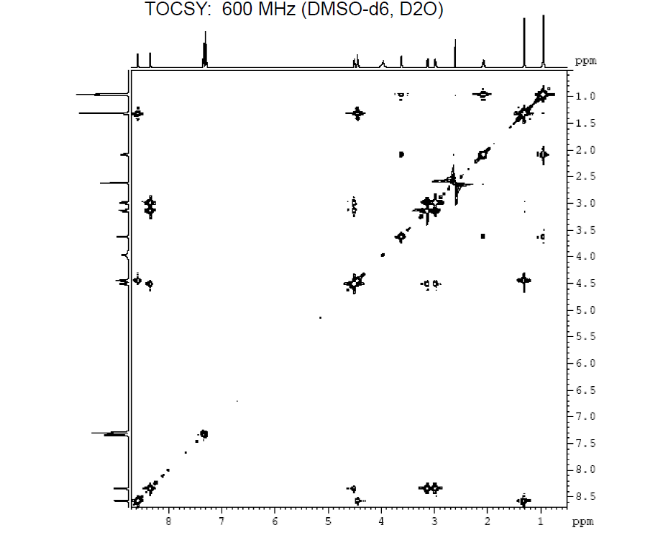
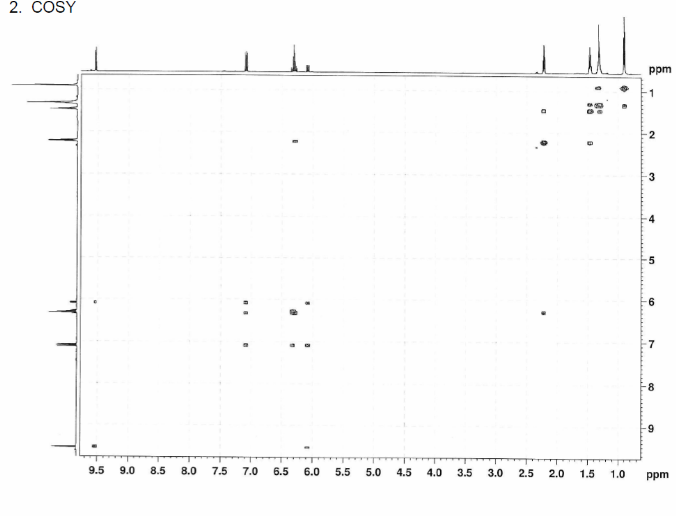
We can also study COSY for finding homonuclear , through bond correlations and (there are other methods also to find heteronuclear through bond correlations)
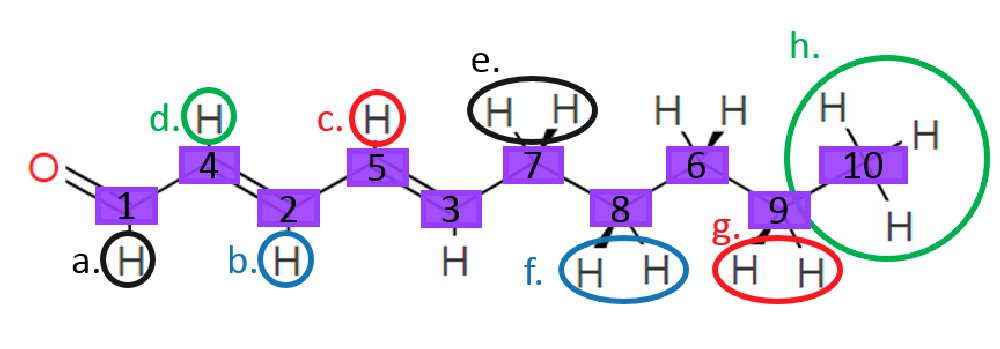
The structure is confirmed to be double conjugated linear aldehyde.

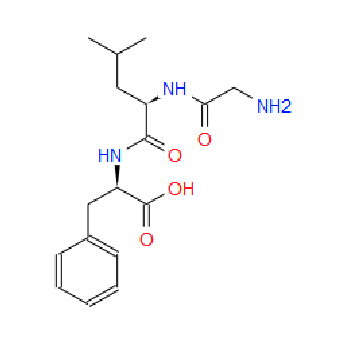
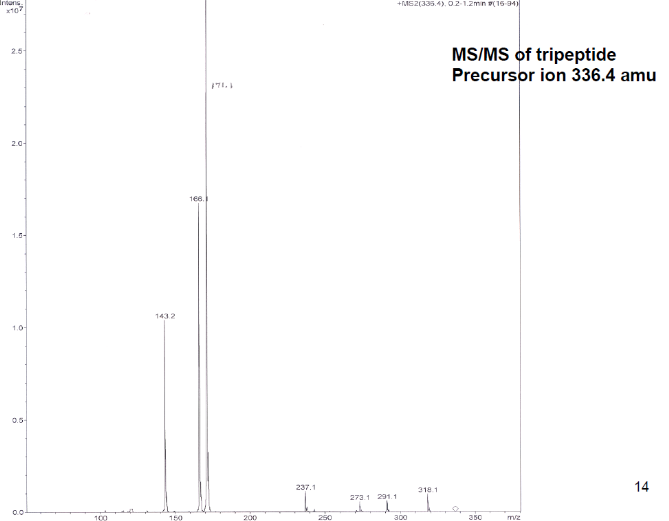
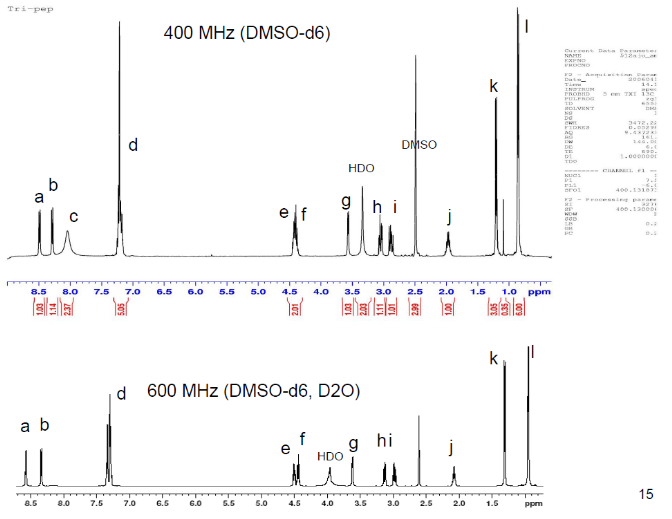
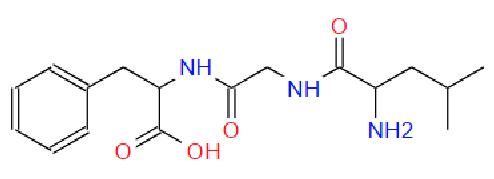
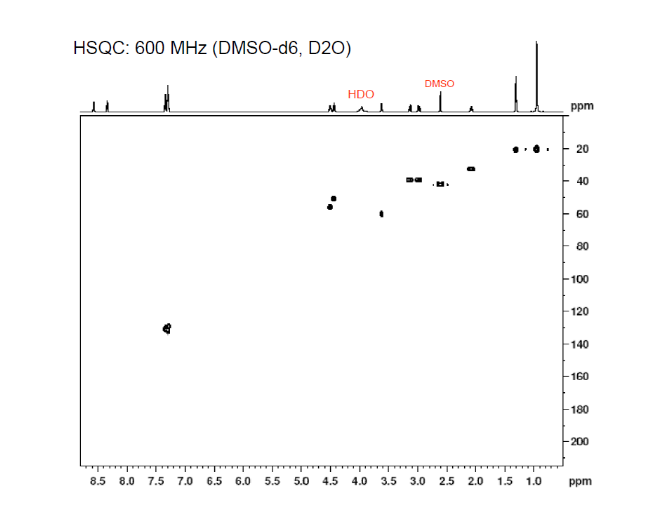
3c. Protons are major factors involved in H-bonding in NMR, which provides the shielding and deshielding effects when we compare the spectra of (2S)-2-[[(2S)-2-[(2-aminoacetyl)amino]-4-methylpentanoyl]amino]-3-phenylpropanoic acid i.e. H-Gly-Leu-Phe-OH at 400 MHz and 600 MHz, we are basically dealing with -OH bonding which can be identified by carrying out a simple D2O (deuterium oxide) which is been added to 600MHz to DMSO. The reaction proceeds as R-OH + D2O à R-OD + HOD during the hydrogen bonding, the -H from alcohol group is exchanged with -D of deuterium so the alcohol becomes R-OD, hence the peak due to -OH disappears and the formation of HOD peak appears. Which is clearly seen the peak “C” in the graph of 400MHz disappears form 600MHz after addition of D2O similarly the generation of new peak of HOD at 4.0 ppm and reduced integration of 3.3ppm peak of 400MHz